- Center on Health Equity and Access
- Clinical
- Health Care Cost
- Health Care Delivery
- Insurance
- Policy
- Technology
- Value-Based Care
Most US Dermatologists Prescribe Baricitinib for Alopecia Areata, Report Shows
Baricitinib is more commonly prescribed by dermatologists for patients with alopecia in the United States and is projected to maintain a substantial market lead over ritlecitinib.
This article was originally published by Dermatology Times®. This version is lightly edited.
Man with bald patches
Image credit: alex papp - adobe.stock.com
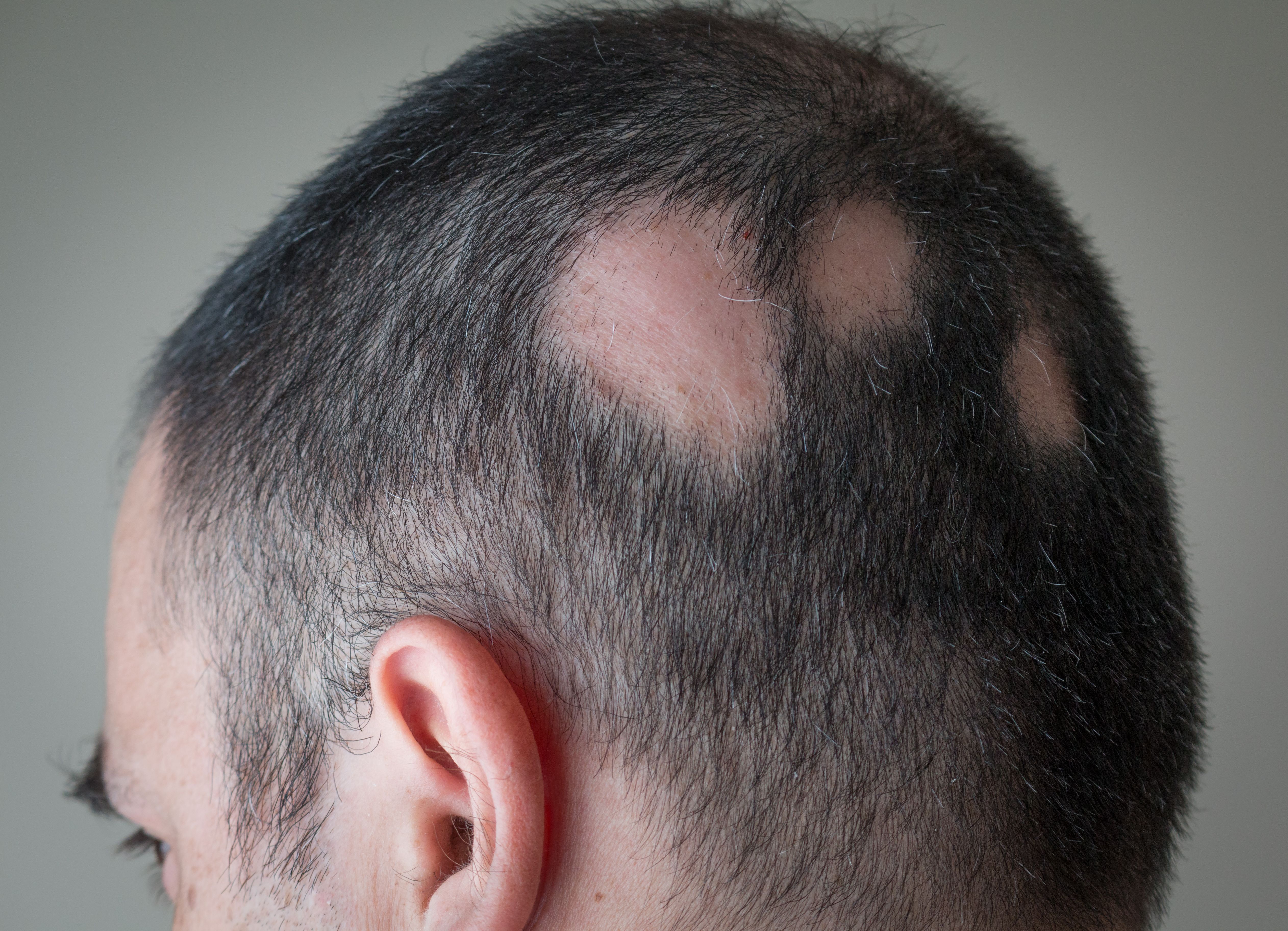
Bariticinib (Olumiant; Eli Lilly) is prescribed by a majority of US dermatologists to their patients with alopecia areata (AA), according to a recent analysis. Additionally, more than half of US dermatologists have begun trials of ritlecitinib (Litfulo; Pfizer) in their patients with AA.
According to the analysis, 6-month market projections for both drugs are favorable. However, bariticinib is expected to maintain a substantial market lead over ritlecitinib.
Spherix Global Insights notes that within the past 4 years of their analysis of the AA market, the introduction of Janus kinase (JAK) inhibitors has made a significant impact on the market itself, particularly attributable to its ability to meet an unmet need in severe alopecia.
The analysis found that 105 US dermatologists currently prescribe baricitinib to their patients with AA, potentially attributable to the JAK inhibitor's earlier introduction in the space for AA management.
A specific analysis of 520 charts of patients who have AA reaffirmed this prescription rate, with prescribers noting the choice to prescribe baricitinib could mostly be attributable to familiarity and comfort. The most common factor for initiating treatment with ritlecitinib, the analysis found, was a prescriber and patient desire to trial a newer therapy.
Despite the difference in the number of dermatologists prescribing bariticinib vs ritlecitinib for patients with AA, the analysis found that prescribers of both drugs expressed comparable and high levels of satisfaction. In fact, these rates of satisfaction were significantly greater than rates for nonadvanced systemic treatments.
Both drugs received equally favorable evaluations of overall patient satisfaction, eyebrow and eyelash regrowth, and Severity of Alopecia Tool scoring.
"Recent data from Spherix's published studies highlight minimal differentiation among prescribers regarding the 2 JAK inhibitors," according to Spherix. "A significant portion—two-fifths—explicitly state no differentiation between the two, while one-quarter of the remaining respondents emphasize that dosing represents the most notable contrast. Fewer than one-fifth report differences in access and efficacy between the brands."
The analysis also found that for manufacturers of both drugs, "Dermatologists expressed a shared need for support in access/improvement of patient assistance programs, increased provision of samples, enhanced resources for patient and physician education, and an overall demand for more comprehensive data."
Despite these expressed needs, Spherix found that prescribers were generally unaware of the AA drug pipeline. Most prescribers involved in the analysis were not able to recall other AA treatments aside from JAK inhibitors in development, but were able to recall well-known line-extensions such as upadacitinib (Rinvoq; AbbVie) and deucravacitinib (Sotyktu; BMS) when prompted.
The top drugs currently in development, ranked by prescribers based on familiarity, included deuruxolitinib (Sun Pharmaceutical Industries), etrasimod (Velsipity; Pfizer), bempikibart (Q32 Bio andAmgen/Horizon), and ivarmacitinib (Arcutis).
Less familiar to prescribers were 3 novel therapies currently in development: daxdilimab (Amgen/Horizon), farudostat (ASLAN Pharmaceuticals), and EQ101 (Equillium).
Reference
Eli Lilly’s Olumiant and Pfizer’s Litfulo strengthen alopecia areata arsenal, yet resounding opportunity for JAK expansion and new assets in development remain. GlobeNewswire. Spherix Global Insights. November 15, 2023. Accessed November 16, 2023. https://www.globenewswire.com/news-release/2023/11/15/2781240/0/en/Eli-Lilly-s-Olumiant-and-Pfizer-s-Litfulo-Strengthen-Alopecia-Areata-Arsenal-Yet-Resounding-Opportunity-for-JAK-Expansion-and-New-Assets-in-Development-Remain.html
Real-World Data Show Sotorasib Effective for NSCLC With KRAS Mutation
May 18th 2024Data from real-world and clinical-trial settings on frontline monotherapy treatment with the KRAS inhibitor sotorasib both show similar progression-free survivals and a high likelihood that the treatment’s efficacy is not affected with dose reduction.
Read More
Health Equity and Access Weekly Roundup: May 18, 2024
May 18th 2024The US Senate hosted a panel addressing physician and health care shortages and efforts to increase minority representation in the medical field. An expert discussed initiatives to prevent senior homelessness. Advocates called for the repeal of the Comstock Act. Regulatory reforms are called for to improve rural cancer patients' access to pharmacies. Research reveals the impact of denials on patient access to immunology treatments.
Read More
Frameworks for Advancing Health Equity: Urban Health Outreach
May 9th 2024In the series debut episode of "Frameworks for Advancing Health Equity," Mary Sligh, CRNP, and Chelsea Chappars, of Allegheny Health Network, explain how the Urban Health Outreach program aims to improve health equity for individuals experiencing homelessness.
Listen
