- Center on Health Equity and Access
- Clinical
- Health Care Cost
- Health Care Delivery
- Insurance
- Policy
- Technology
- Value-Based Care
Amgen Announces $19M in Q2 Revenue From Amjevita; Humira Down 26% in US
Amjevita’s revenue was down 63% in the second quarter (Q2) compared with the first quarter of 2023, when the biosimilar launched.
For half a year, Amjevita, Amgen’s adalimumab biosimilar, had the US market all to itself after it launched January 31, 2023. However, in the second quarter (Q2), the biosimilar only brought in $19 million in revenue.1
The reference product, Humira, still had net revenues in the United States of $3.452 billion.2
Globally, sales for Amjevita and Amgevita—what the biosimilar is marketed as outside of the United States—had increased 29% year over year. Total global sales in Q2 were $150 million. Sales outside of the United States were $131 million in Q2 2023, up 12.9% over a year ago.
Sales in the United States were down 63% quarter over quarter driven by a drawdown in inventory levels following the inventory build to support the product’s launch, according to an Amgen press release.1
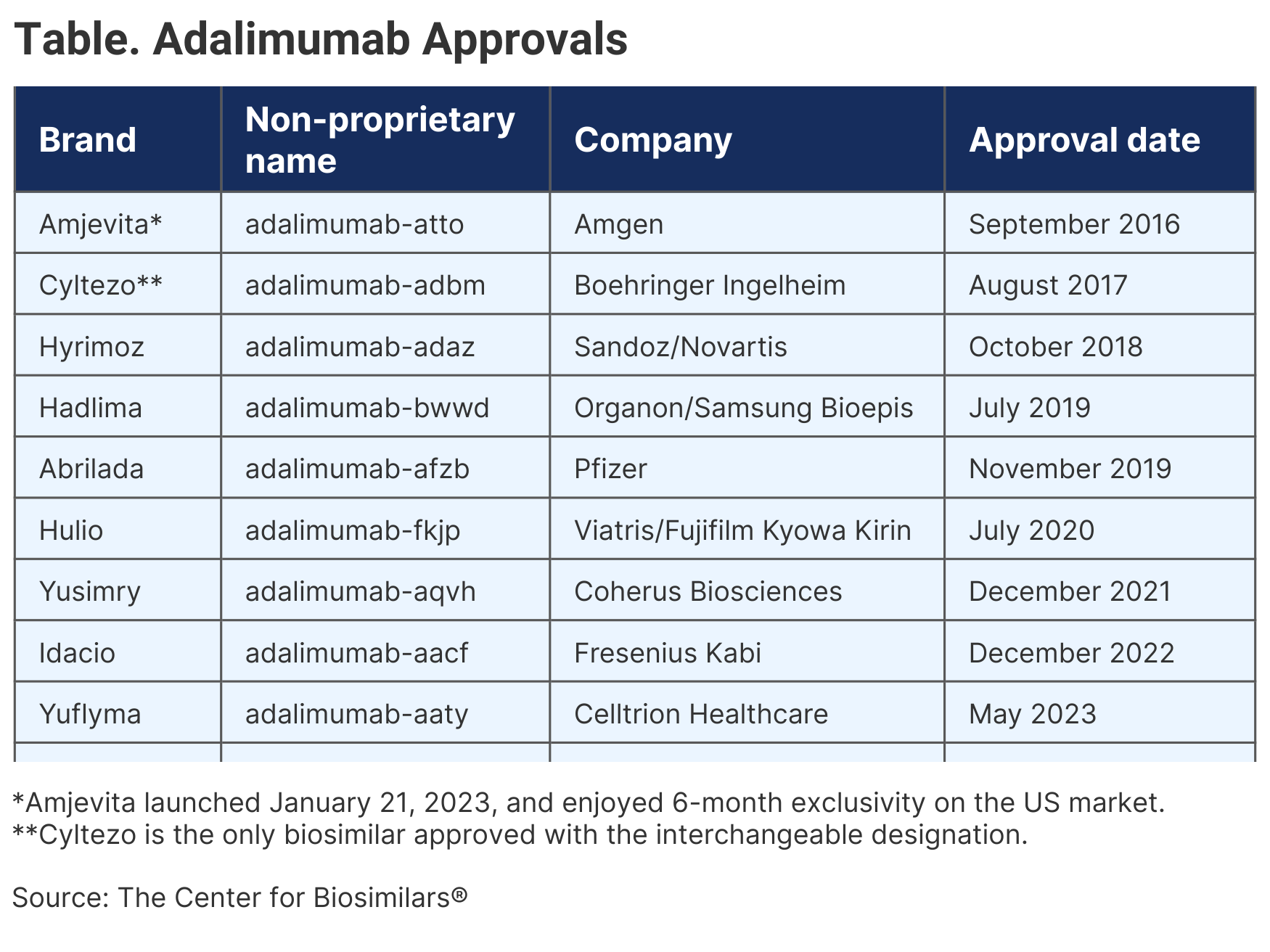
In a year with as many as 9 adalimumab biosimilars expected to launch in the United States, Amjevita held a coveted spot because it had market exclusivity for 6 months before more competition hit (Table). And more competitors may be coming.
Teva Pharmaceuticals is pursuing its own adalimumab biosimilar with an eye on a 2024 launch as long as the company’s Iceland production site passes an inspection. Teva’s CEO, Richard Francis, told Reuters that despite the challenging market, he still sees launching another adalimumab as being worthwhile because the US biosimilars market is still evolving.3
After Amjevita, another 7 biosimilars launched. Currently, the only approved adalimumab biosimilar that has yet to launch is Abrilada from Pfizer. Among the competitors now on the market is Cyltezo, the only adalimumab biosimilar that has so far been granted interchangeability, which some experts expect will give Cyltezo an advantage because it can be substituted at the pharmacy.
A report from Cardinal Health found that more than 60% of surveyed providers only want to prescribe adalimumab biosimilars with the interchangeability designation.4 While Cyltezo is currently the only adalimumab biosimilar with the designation, Hadlima, Abrilada, and Yuflyma are expected to seek the designation. In addition, Alvotech is submitting its candidate, AVT02, to the FDA for approval as an interchangeable adalimumab biosimilar.
At the end of July, AbbVie, the maker of Humira, announced second-quarter financial results, including that global Humira net revenues were just over $4 billion, a decrease of 25.2% on a reported basis compared with the same quarter a year ago. In the United States, Humira’s net revenues were down 26.0%.
In comparison, risankizumab (Skyrizi) and upadacitinib (Rinvoq) global net revenues were up 50.4% and 55.1%, respectively, on a reported basis.
Richard A. Gonzalez, chair and CEO of AbbVie, noted that the company’s performance was driven “predominantly by our non-Humira business.” However, he noted in a conference call that the company is pleased with how well Humira is competing against biosimilar competition so far, reported FiercePharma.5
Amgen also reported sales decreases for 2 other biosimilars, bevacizumab-awwb (Mvasi) and trastuzumab-anns (Kanjinti). Year over year, Mvasi’s sales were down 19% and Kanjinti’s sales were down 41%, both driven by lower net selling. “Going forward, we expect continued net selling price erosion,” Amgen said about both products,1 driven by increased competition.
References
1. Amgen reports second quarter financial results. Press release. Amgen. August 3, 2023. Accessed August 7, 2023. https://www.amgen.com/newsroom/press-releases/2023/08/amgen-reports-second-quarter-financial-results
2. AbbVie reports second-quarter 2023 financial results. Press release. AbbVie. July 27, 2023. Accessed August 7, 2023. https://news.abbvie.com/news/press-releases/abbvie-reports-second-quarter-2023-financial-results.htm
3. Scheer S. Teva CEO says not too late to launch Humira biosimilar in 2024. Reuters. August 2, 2023. Accessed August 7, 2023. https://www.reuters.com/business/healthcare-pharmaceuticals/teva-pharm-ceo-not-too-late-launch-humira-biosimilar-2024-2023-08-02/
4. Jeremias S. Report: over 60% of providers plan to only prescribe interchangeable adalimumab biosimilars. The Center for Biosimilars®. February 23, 2023. Accessed August 7, 2023. https://www.centerforbiosimilars.com/view/report-over-60-of-providers-plan-to-only-prescribe-interchangeable-adalimumab-biosimilars
5. Becker Z. Despite big Q2 declines for Humira and Imbruvica, AbbVie still boosts earnings guidance. FiercePharma. July 27, 2023. Accessed August 7, 2023. https://www.fiercepharma.com/pharma/abbvie-optimistic-halfway-point-humiras-year-biosimilar-attacks-lowers-estimate-biosimilar
Frameworks for Advancing Health Equity: Urban Health Outreach
May 9th 2024In the series debut episode of "Frameworks for Advancing Health Equity," Mary Sligh, CRNP, and Chelsea Chappars, of Allegheny Health Network, explain how the Urban Health Outreach program aims to improve health equity for individuals experiencing homelessness.
Listen
Study Highlights Significant Increases in Utilization, Spending on DMD Drugs in Medicaid
May 17th 2024The findings add to recent research on the growing utilization, expenditure, and prices of Duchenne muscular dystrophy (DMD) therapies in the current landscape, an area health care policy could potentially address.
Read More
Oncology Onward: A Conversation With Thyme Care CEO and Cofounder Robin Shah
October 2nd 2023Robin Shah, CEO of Thyme Care, which he founded in 2020 with Bobby Green, MD, president and chief medical officer, joins hosts Emeline Aviki, MD, MBA, and Stephen Schleicher, MD, MBA, to discuss his evolution as an entrepreneur in oncology care innovation and his goal of positively changing how patients experience the cancer system.
Listen
Panel Addresses Minority Physician Shortage, Maternal Health at Senate Committee Hearing
May 15th 2024The senate hearing held by the US Senate Committee on Health, Education, Labor, and Pensions, led by Sen. Bernie Sanders (I, Vermont), chairman of the committee, and ranking member Sen. Bill Cassidy, MD, (R, Louisiana), addressed the critical issue of physician and health care worker shortages, as well as the maternal health crisis, in the US.
Read More
Bleeds and Resource Use in Hemophilia B: Retrospective Observational Study
May 15th 2024This real-world US study describes individuals with hemophilia B who experience bleeds despite factor replacement therapy and quantifies the associated comorbidity and health care resource utilization burden.
Read More
